Homogenized Implant Sterile, Biodegradable Good Biocompatibility Cross-Linked Sodium Hyaluronate Gel with CE Approved
XIAMEN POPTRIMS TEXTILE CO.,LTD. / 2022-06-23
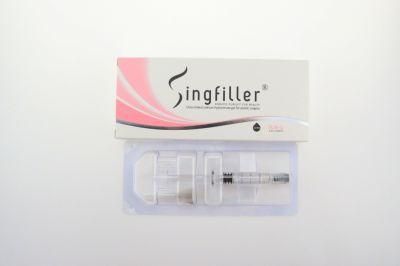
- Certification:FDA, QS, CCC, RoHS, ISO, CE, Sfda
- Disinfection:Without Disinfection
- Color:Transparent
- Application:Face Wrinkle
- Material:Hyaluronic Acid
- Supply Tyle:OEM, Obl, ODM
=== Base Info ===
- Model NO.:singfiller deeper
- Suitable for:Adults
- Particle Size:0.28~0.5mm
- Duration Time:6~12 Months
- Storage:up to 30 Degree
- Syringe:Bd 1.0 Prtc
- Form:Gel
- Function:Anti-Aging
- Gender:Unisex
- Shelf Life:2 Years
- Transport Package:Prefilled Syringe in Blister
- Specification:20mg,ml, 1ml
- Trademark:Singfiller
- Origin:China
- HS Code:3913900090
- Production Capacity:100000PCS,Year
=== Description ===
Overview
Product Description
Main Products



New product
Hot product
- Gm Candi Interface
2022-06-23
- Scan Dash V2.0 for BMW
2022-06-23
- K+Can Commander 2.0
2022-06-23
- Mut-3 Professional Diagnostic Tool Mut3
2022-06-23
- CKM-100 Key Programmer for Benz/BMW
2022-06-23
- Fuel Injector Tester & Cleaner (MST-N6A)
2022-06-23
- Mst 9000+ Plus ECU Simulator
2022-06-23
- OBD Ii Code Reader Mst-300, OBD2 Code Scanner
2022-06-23
- Digital Battery Analyzer (SC-100)
2022-06-23
- VAS5052A PC VERSION with VAS5054A Wireless Bluetooth Communicate
2022-06-23





